Press
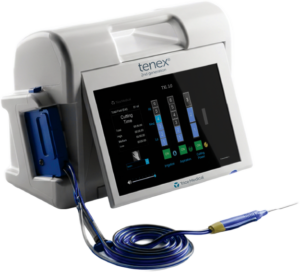
TRICE MEDICAL LAUNCHES TENEX® 2ND GENERATION ULTRASONIC SYSTEM, EXPANDING MINIMALLY INVASIVE TREATMENT FOR CHRONIC TENDON PAIN
Next-generation platform now offers deeper tissue access and enhanced procedural efficiency for physicians Malvern, PA (March 17, 2026) – Trice Medical today announced nationwide commercial availability

Tenex® 2nd Generation: FDA Clears the Next Innovation in Tenex Technology
Tenex® 2nd Generation: FDA Clears the Next Innovation in Tenex Technology Malvern, PA (December 4, 2024) – Trice Medical has received FDA 510(k) Clearance for

Trice Medical breaks surgical boundaries with the world’s first FDA-cleared single-use 25° needle arthroscope – giving surgeons the tools they need to see more pathology
Malvern, PA (October 26, 2021) – Trice Medical has received FDA 510(k) Clearance for 25-degree mi-eye 3 needlescope™ with distinctive horizon leveling technology, clearing the way for

Trice Medical Receives Investment and International Distribution Partnership from Bioventus, Inc.
Malvern, PA (August 26th, 2021) – Trice Medical announced today that it received a strategic investment from Bioventus Inc. (Nasdaq: BVS), a global leader in innovations for

Ryan Hartman Appointed to Chief Commercial Officer of Trice Medical
Malvern, PA (May 24th, 2021) – Trice Medical announced today the appointment of Ryan Hartman as Chief Commercial Officer. Mr. Hartman brings with him over

Trice Medical acquires Tenex Health
Trice Medical continues mission of making elective procedures more efficient through the acquisition of Tenex Health. Malvern, PA (April 1st, 2021) – Trice Medical announced
