Revolutionizing how you heal diabetic foot ulcers.
Treat a root of the problem, not just the surface.
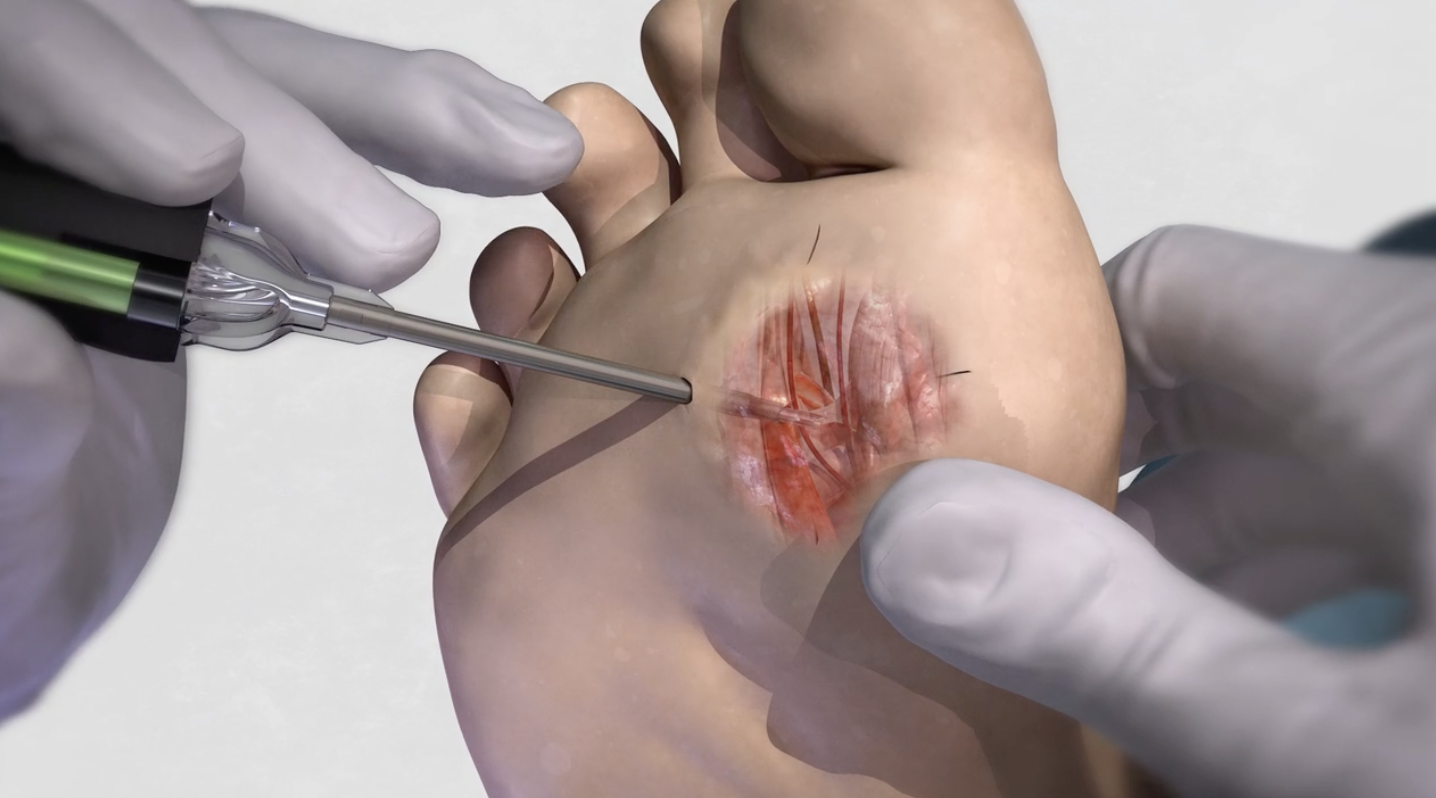
The Tenex Health TX® System helps heal diabetic foot ulcers in a single treatment. It allows physicians safe access under the wound – to relieve pressure ulcers by removing the bony prominence and unhealthy tissue. This is accomplished without entering or expanding the wound area. It is an out-patient procedure that takes minutes to perform.
The Tenex Health TX® System and TX-Bone MicroTip are FDA cleared for treating diabetic foot ulcers. The clinical experience in a study of 105 patients show 96% of patients healed in less than 3.5 weeks on average and a recurrence rate of less than 5%.†
Explore Tenex for Treating DFU
DFU treatment and benefits
Technology, benefits, and treatment pathway
Tenex vs. Conventional DFU treatments
Diabetic Foot Ulcer Procedure Demo
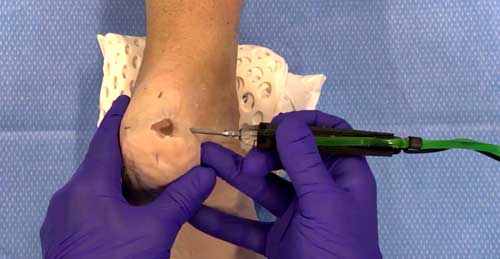
Specification
| Image | Title | Categories | Link | File Type | hf:doc_categories |
|---|---|---|---|---|---|
| Technique Guide DFU – MKT-315 REV E | Technique Guide | technique-guide | |||
| Patient Brochure DFU – MKT-389 Rev B | Brochure | brochure | |||
| Sales Sheet – Podiatry – MKT-297 Rev F | Brochure | brochure | |||
| DFU Patient Microsite MKT-385 Rev A | Website | www | website |